
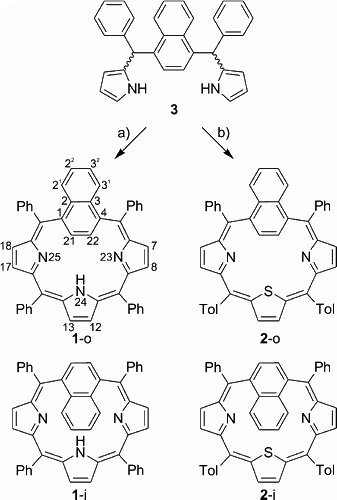
The carbaporphyrinoids 5,10,15,20-tetraphenyl-1,4-naphthiporphyrin and 5,20-diphenyl-10,15-ditolyl-24-thia-1,4-naphthiporphyrin, which contain a naphthalene ring embedded in the macrocyclic framework, were obtained by B. Szyszko in a modification of the “3 + 1” approach using the naphthalene analogue of tripyrrane.
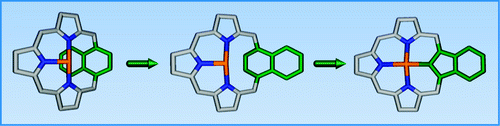
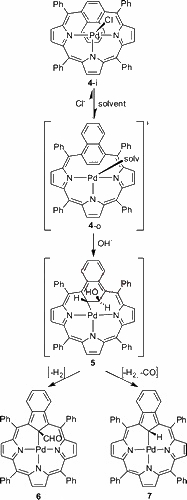
The reaction of palladium(II) chloride with 1,4-naphthiporphyrin in acetonitrile yielded palladium(II) 1,4-naphthiporphyrin, which preserved the folded conformation of the free base. The remarkable, facile palladium(II)-stimulated contraction of naphthalene to isoindene produced the first complex of metal(II) benzocarbaporphyrin which acquires a unique tetrahedral carbon coordination mode.
Further reading
Szyszko, B.; Latos-Grażyński, L. Conformational Flexibility of 1,4-Naphthiporphyrin Promotes a Palladium-Mediated Contraction of Naphthalene to Isoindene Organometallics 2011, 30, 4354.