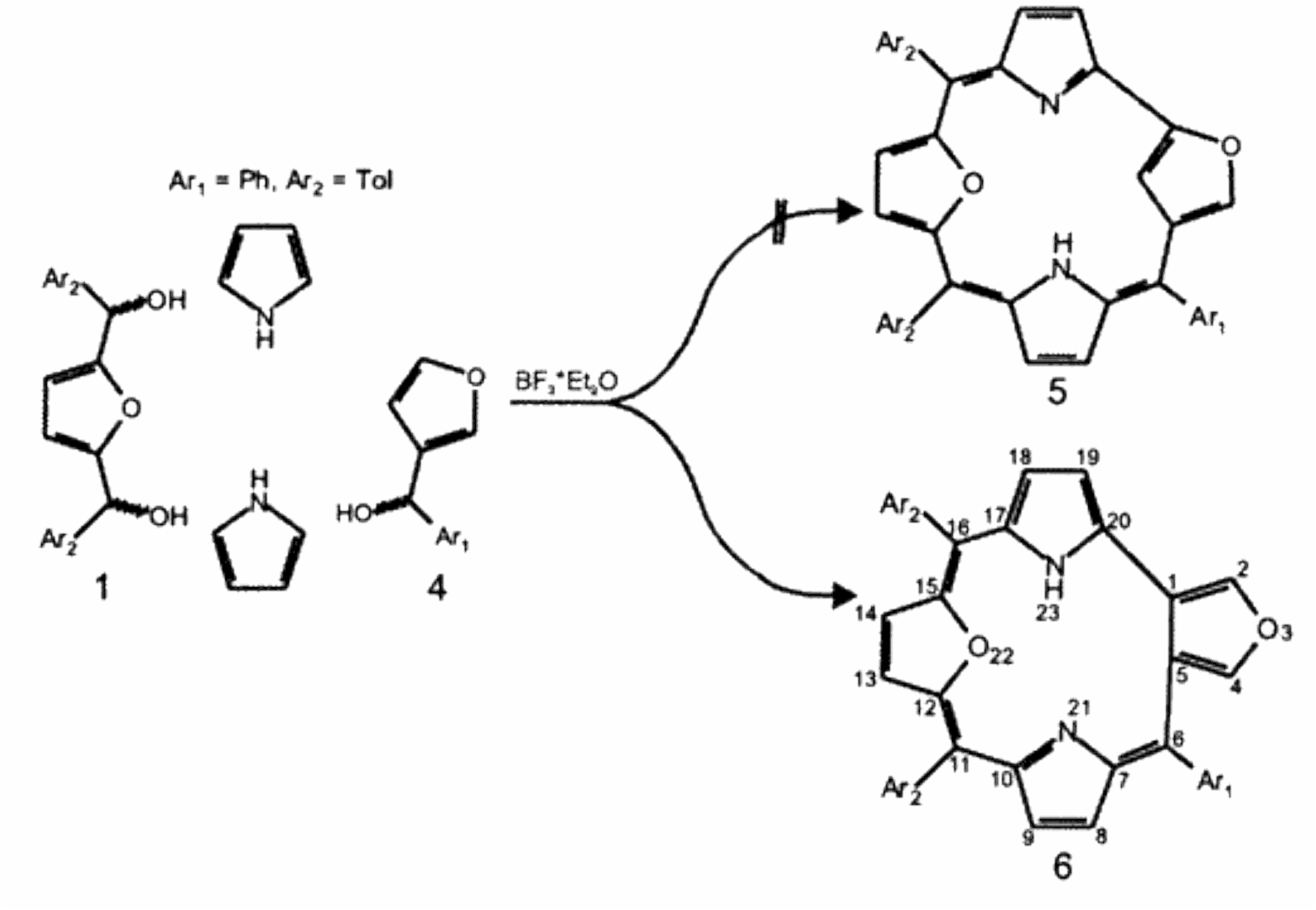
M. Pawlicki, A Kędzia et al. conducted investigation of oxatriphyrin(3.1.1) properties. An oxa-analogue of 5,10,15-triarylcorrole, i.e., 5,10,15-triaryl-21,23-dioxacorrole (21,23-O2Cor)H, where two pyrrole rings are replaced by furan moieties, has been produced by condensation of 2,5-bis(arylhydroxymethyl)furan, 2-phenylhydroxymethylfuran, and pyrrole.

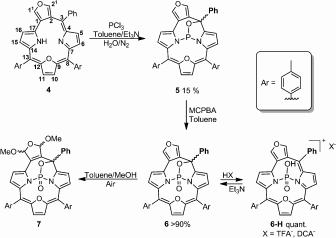
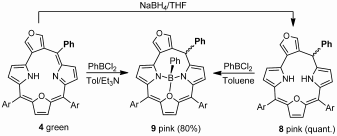
Furan-fused oxatriphyrin(3.1.1) acts as a ligand towards boron(III), phosphorous(III), and phosphorous(V) ions. The coordination abilities are adjusted by changes of the oxidation state.
Further reading
Pawlicki, M.; Kędzia, A.; Bykowski, D.; Latos-Grażyński, L. Reversible Reduction of Oxatriphyrin(3.1.1)—Adjusting the Coordination Abilities to the Central Ion. Chem. Eur. J. 2014, 20, 17500.
5,10,15-Triaryl-21,23-dioxacorrole and Its Isomer with a Protruding Furan Ring. J. Org. Chem. 2002, 67, 5644.