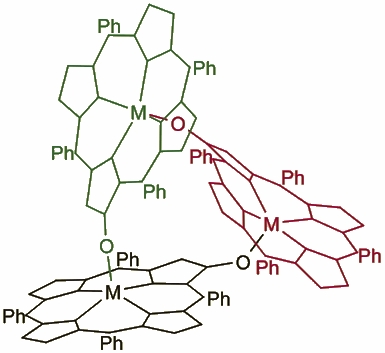

Iron b-Hydroxyporphyrin trimer was synthesized by J.Wojaczynski. The 1H NMR spectroscopic and mass spectrometric investigations indicate that these compounds have a head-to-tail cyclic trimeric structure. In the 1H NMR spectra the interactions between paramagnetic, weakly coupled centers are reflected by marked variations of chemical shifts and line widths of pyrrole resonances. The characteristic upfield positions of the 3-H pyrrole resonances are diagnostic for the trimeric motifs. The structure of the prototypical molecule, [(2-O-TTP)FeIII]3, has been determined by X-ray crystallography.The trimeric iron(III)complex has a head-to-tail cyclic arrangement with the pyrrolic alkoxide groups formingbridges from one macrocycle to the metal in adjacent macrocycle. The three iron(III) porphyrin subunits arenot equivalent but have typical geometry for high-spin five-coordinate iron(III) porphyrin complexes. The porphyrin skeleton of [(2-O-TTP)FeIII]3 is expected to be representative of the structures of the homometallic and heterometallic trimeric complexes of 2-hydroxytetraarylporphyrin with M(III) ions.
Further reading
J. Wojaczyński, L. Latos-Grażyński, Synthesis and Characterization of High-Spin Iron(III) 2-Hydroxy-5,10,15,20-tetraphenylporphyrin. The Unprecendented Example of the Cyclic Iron Porphyrin Trimer. Inorg. Chem., 1995, 34, 1044-1053.
J. Wojaczyński, L. Latos-Grażyński, Synthesis and Characterization of Gallium(III) 2-Hydroxy-5,10,15,20-tetraphenylporphyrin. A Novel Example of a Cyclic Gallium Porphyrin Trimer. Inorg. Chem., 1995, 34, 1054-1062.
J. Wojaczyński, L. Latos-Grażyński, Monomeric and Trimeric Manganese(III) Complexes of 2-Hydroxy-5,10,15,20-tetraphenylporphyrin. Synthesis and Characterization.Inorg. Chem., 1996, 35, 4812-4818.
J. Wojaczyński, L. Latos-Grażyński, M. M. Olmestead, A. L. Balch, Cyclic Metalloporphyrin Trimers : 1H NMR Identification of Trimeric Heterometallic (Iron(III), Gallium(III), Manganese(III))2-Hydroxy-5,10,15,20-tetraphenylporphyrins and X-Ray Crystal Structure of Iron(III) 2-Hydroxy-5,10,15,20-tetra-p-tolyl-porphyrin Trimer. Inorg. Chem., 1997, 36, 4548-4554.
(See the complete publication list of our group.)